Page 21 - EASL Recommendations on Treatment of Hepatitis C 2015 - Summary
P. 21
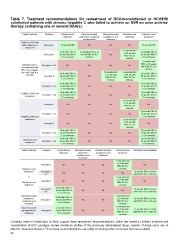
Table 7. Treatment recommendations for retreatment of HCV-monoinfected or HCV/HIV
coinfected patients with chronic hepatitis C who failed to achieve an SVR on prior antiviral
therapy containing one or several DAA(s).
Failed treatment Genotype Sofosbuvir and Ritonavir-boosted Ritonavir-boosted Sofosbuvir and Sofosbuvir and
Genotype 1 ledipasvir paritaprevir, ombitasvir paritaprevir, and simeprevir daclatasvir
PegIFN-α, RBV and Genotype 1
either telaprevir or 12 wk with RBV and dasabuvir ombitasvir No 12 wk with RBV
boceprevir No No 12 wk with RBV 12 wk with RBV or
or 24 wk with 24 wk with RBV if
12 wk with RBV or 12 wk with RBV or 24 No RBV if F3 or
24 wk with RBV if wk with RBV if F3 or cirrhosis F3 or cirrhosis
F3 or cirrhosis cirrhosis No 12 weeks with
RBV or 24 weeks
Sofosbuvir alone, Genotype 2 or 3 No No No 12 wk with RBV with RBV if F3 or
in combination with Genotype 4 No or 24 wk with
RBV or in combina- 12 wk with RBV or No 12 wk with RBV RBV if F3 or cirrhosis
tion with PegIFN-α 24 wk with RBV if No or 24 wk with cirrhosis
No RBV if F3 or 12 wk with RBV or
and RBV F3 or cirrhosis cirrhosis No 24 wk with RBV if
No
PegIFN-α, RBV and Genotype 5 or 6 12 wk with RBV or No F3 or cirrhosis
simeprevir Genotype 1 or 4 24 wk with RBV if No
12 wk with RBV 12 wk with RBV or
F3 or cirrhosis No or 24 wk with 24 wk with RBV if
RBV if F3 or
12 wk with RBV or cirrhosis F3 or cirrhosis
24 wk with RBV if 12 wk with RBV or
No 24 wk with RBV if
F3 or cirrhosis
12 wk with RBV F3 or cirrhosis
Genotype 1 No or 24 wk with
RBV if F3 or No
PegIFN-α, RBV and Genotype 2 or 3 No No No cirrhosis
daclatasvir Genotype 4 No No No 12 wk with RBV or
No No No 24 wk with RBV if
Sofosbuvir and Genotype 5 or 6 12 wk with RBV or No No
simeprevir Genotype 1 or 4 24 wk with RBV if No F3 or cirrhosis
F3 or cirrhosis No
12 wk with RBV or 12 wk with RBV or
24 wk with RBV if 24 wk with RBV if
F3 or cirrhosis F3 or cirrhosis
12 wk with RBV or
24 wk with RBV if
F3 or cirrhosis
Failed treatment Genotype Sofosbuvir and Ritonavir-boosted Ritonavir-boosted Sofosbuvir and Sofosbuvir and daclatasvir
ledipasvir paritaprevir, paritaprevir, and simeprevir
Sofosbuvir and Genotype 1
daclatasvir Genotype 2 ombitasvir and ombitasvir
or dasabuvir
or 3 No
Sofosbuvir and Genotype 4 No No No 12 wk with RBV No
ledipasvir Genotype 5 No or 24 wk with
No No RBV if F3 or 12 wk with RBV or 24 wk
Ritonavir-boosted or 6 No cirrhosis with RBV if F3 or cirrhosis
paritaprevir, Genotype 1 No No
No No No No
ombitasvir and Genotype 4 12 wk with RBV or No
dasabuvir 24 wk with RBV if No No 12 wk with RBV 12 wk with RBV or 24 wk
or 24 wk with with RBV if F3 or cirrhosis
Ritonavir-boosted F3 or cirrhosis RBV if F3 or
paritaprevir and cirrhosis 12 wk with RBV or 24 wk
12 wk with RBV or with RBV if F3 or cirrhosis
ombitasvir 24 wk with RBV if No
12 wk with RBV or 24 wk
F3 or cirrhosis 12 wk with RBV with RBV if F3 or cirrhosis
or 24 wk with
12 wk with RBV or RBV if F3 or
24 wk with RBV if cirrhosis
F3 or cirrhosis 12 wk with RBV
or 24 wk with
RBV if F3 or
cirrhosis
Currently, there is limited data to firmly support these retreatment recommendations, which are based on indirect evidence and
consideration of HCV genotype, known resistance profiles of the previously administered drugs, number of drugs used, use of
ribavirin, treatment duration. Thus, these recommendations are subject to change when more data become available.
20
coinfected patients with chronic hepatitis C who failed to achieve an SVR on prior antiviral
therapy containing one or several DAA(s).
Failed treatment Genotype Sofosbuvir and Ritonavir-boosted Ritonavir-boosted Sofosbuvir and Sofosbuvir and
Genotype 1 ledipasvir paritaprevir, ombitasvir paritaprevir, and simeprevir daclatasvir
PegIFN-α, RBV and Genotype 1
either telaprevir or 12 wk with RBV and dasabuvir ombitasvir No 12 wk with RBV
boceprevir No No 12 wk with RBV 12 wk with RBV or
or 24 wk with 24 wk with RBV if
12 wk with RBV or 12 wk with RBV or 24 No RBV if F3 or
24 wk with RBV if wk with RBV if F3 or cirrhosis F3 or cirrhosis
F3 or cirrhosis cirrhosis No 12 weeks with
RBV or 24 weeks
Sofosbuvir alone, Genotype 2 or 3 No No No 12 wk with RBV with RBV if F3 or
in combination with Genotype 4 No or 24 wk with
RBV or in combina- 12 wk with RBV or No 12 wk with RBV RBV if F3 or cirrhosis
tion with PegIFN-α 24 wk with RBV if No or 24 wk with cirrhosis
No RBV if F3 or 12 wk with RBV or
and RBV F3 or cirrhosis cirrhosis No 24 wk with RBV if
No
PegIFN-α, RBV and Genotype 5 or 6 12 wk with RBV or No F3 or cirrhosis
simeprevir Genotype 1 or 4 24 wk with RBV if No
12 wk with RBV 12 wk with RBV or
F3 or cirrhosis No or 24 wk with 24 wk with RBV if
RBV if F3 or
12 wk with RBV or cirrhosis F3 or cirrhosis
24 wk with RBV if 12 wk with RBV or
No 24 wk with RBV if
F3 or cirrhosis
12 wk with RBV F3 or cirrhosis
Genotype 1 No or 24 wk with
RBV if F3 or No
PegIFN-α, RBV and Genotype 2 or 3 No No No cirrhosis
daclatasvir Genotype 4 No No No 12 wk with RBV or
No No No 24 wk with RBV if
Sofosbuvir and Genotype 5 or 6 12 wk with RBV or No No
simeprevir Genotype 1 or 4 24 wk with RBV if No F3 or cirrhosis
F3 or cirrhosis No
12 wk with RBV or 12 wk with RBV or
24 wk with RBV if 24 wk with RBV if
F3 or cirrhosis F3 or cirrhosis
12 wk with RBV or
24 wk with RBV if
F3 or cirrhosis
Failed treatment Genotype Sofosbuvir and Ritonavir-boosted Ritonavir-boosted Sofosbuvir and Sofosbuvir and daclatasvir
ledipasvir paritaprevir, paritaprevir, and simeprevir
Sofosbuvir and Genotype 1
daclatasvir Genotype 2 ombitasvir and ombitasvir
or dasabuvir
or 3 No
Sofosbuvir and Genotype 4 No No No 12 wk with RBV No
ledipasvir Genotype 5 No or 24 wk with
No No RBV if F3 or 12 wk with RBV or 24 wk
Ritonavir-boosted or 6 No cirrhosis with RBV if F3 or cirrhosis
paritaprevir, Genotype 1 No No
No No No No
ombitasvir and Genotype 4 12 wk with RBV or No
dasabuvir 24 wk with RBV if No No 12 wk with RBV 12 wk with RBV or 24 wk
or 24 wk with with RBV if F3 or cirrhosis
Ritonavir-boosted F3 or cirrhosis RBV if F3 or
paritaprevir and cirrhosis 12 wk with RBV or 24 wk
12 wk with RBV or with RBV if F3 or cirrhosis
ombitasvir 24 wk with RBV if No
12 wk with RBV or 24 wk
F3 or cirrhosis 12 wk with RBV with RBV if F3 or cirrhosis
or 24 wk with
12 wk with RBV or RBV if F3 or
24 wk with RBV if cirrhosis
F3 or cirrhosis 12 wk with RBV
or 24 wk with
RBV if F3 or
cirrhosis
Currently, there is limited data to firmly support these retreatment recommendations, which are based on indirect evidence and
consideration of HCV genotype, known resistance profiles of the previously administered drugs, number of drugs used, use of
ribavirin, treatment duration. Thus, these recommendations are subject to change when more data become available.
20